On the nature and plasticity of cancer stem cell phenotype
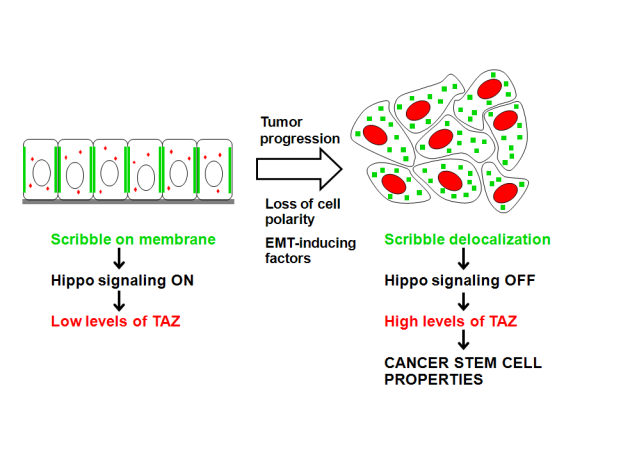
Cancer Stem Cells (CSCs) are proposed to drive tumor initiation and progression. But what is "making" a cancer cell a cancer stem cell? In fact, our understanding of the cellular and molecular mechanisms that underlie CSC phenomenon is limited. We discovered that TAZ, a transducer of the Hippo pathway, sustains self-renewal and tumor-initiation capacities of breast CSCs (Cordenonsi et al., Cell 2011). TAZ protein levels and activity are elevated in prospective CSCs and in poorly differentiated human tumors; and TAZ/YAP activity identifies tumors that will form metastases and with poor prognosis. Gain-of-TAZ endows self-renewal capacity to non-CSCs, and confers them capacity to resist to chemotherapy. TAZ is downstream and mediates the effects of the EMT phenomena, a morphological change known to endow cells with capacities of self-renewal and motility. In sum, we linked the CSC concept to the regulation of the Hippo pathway, and provided a mechanistic basis of the control of Hippo kinases by cell polarity (Cordenonsi et al., Cell 2011). Intriguingly, YAP/TAZ regulation by the Hippo pathway control organ growth during development; thus, finding TAZ as determinant of tumor growth supports the hypothesis that tumors are aberrant attempts at de novo organogenesis.
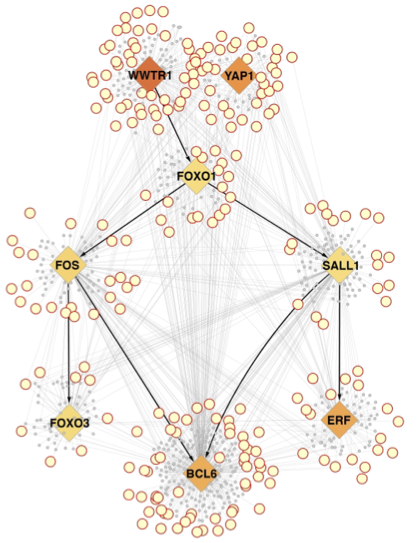
We recently advanced in these studies expanding the functional exploration of YAP/TAZ in non-epithelial tumors. By single-cell analyses we recently characterized the native, endogenous Glioblastoma SCs (GSCs) at the apex of the differentiation hierarchy of glioblastoma, the most frequent and uncurable form of brain cancer. By reconstructing the GSCs' regulatory network, we identified the YAP/TAZ coactivators as master regulators of this cell state. We found that YAP/TAZ install GSC properties in primary, otherwise normal astrocytes, and this occurs downstream of multiple oncogenic lesions. YAP/TAZ are required for GMB initiation and maintenance in vivo in different mouse and human GBM models. In this context, YAP/TAZ act as main roadblock of GSC differentiation, and their inhibition irreversibly locks differentiated GBM cells into a nontumorigenic state, preventing plasticity and regeneration of GSC-like cells. Thus, GSC identity is linked to YAP/TAZ as regulators of the balance between stemness and differentiation, as such serving as integrating hubs of cancer genetics and microenvironmental inputs controlling the multifaceted biology of GBM. See our recent work by Castellan et al. Nature Cancer, 2021.
Click here for a list of protocols on detecting YAP and TAZ proteins by western blot, IHC and IF.